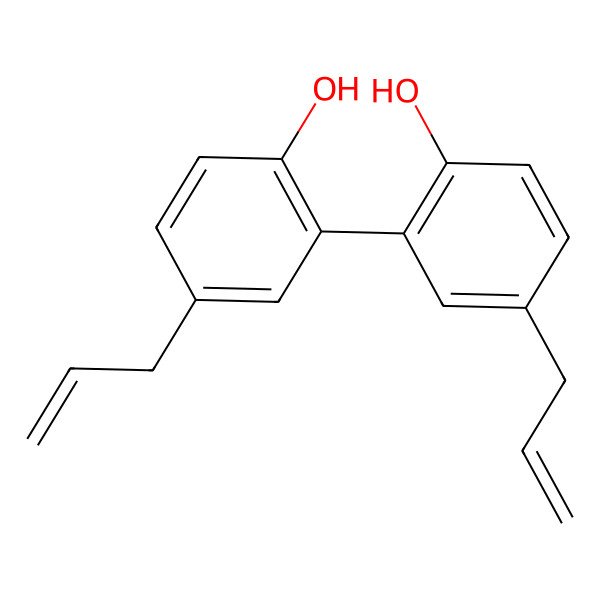
Magnolol
| Internal ID | d4f54e8a-1b51-433b-803f-125f94c8e46b |
| Taxonomy | Benzenoids > Benzene and substituted derivatives > Biphenyls and derivatives |
| IUPAC Name | 2-(2-hydroxy-5-prop-2-enylphenyl)-4-prop-2-enylphenol |
| SMILES (Canonical) | C=CCC1=CC(=C(C=C1)O)C2=C(C=CC(=C2)CC=C)O |
| SMILES (Isomeric) | C=CCC1=CC(=C(C=C1)O)C2=C(C=CC(=C2)CC=C)O |
| InChI | InChI=1S/C18H18O2/c1-3-5-13-7-9-17(19)15(11-13)16-12-14(6-4-2)8-10-18(16)20/h3-4,7-12,19-20H,1-2,5-6H2 |
| InChI Key | VVOAZFWZEDHOOU-UHFFFAOYSA-N |
| Popularity | 1,188 references in papers |
| Molecular Formula | C18H18O2 |
| Molecular Weight | 266.30 g/mol |
| Exact Mass | 266.130679813 g/mol |
| Topological Polar Surface Area (TPSA) | 40.50 Ų |
| XlogP | 5.00 |
| 528-43-8 |
| 5,5'-Diallyl-[1,1'-biphenyl]-2,2'-diol |
| 5,5'-Diallyl-2,2'-biphenyldiol |
| 2,2'-Bichavicol |
| 5,5'-Diallyl-2,2'-dihydroxybiphenyl |
| Dehydrodichavicol |
| NSC 293099 |
| 2-(2-hydroxy-5-prop-2-enylphenyl)-4-prop-2-enylphenol |
| 4-allyl-2-(5-allyl-2-hydroxy-phenyl)phenol |
| MFCD00016658 |
| There are more than 10 synonyms. If you wish to see them all click here. |
| Target | Value | Probability (raw) | Probability (%) |
|---|---|---|---|
| No predicted properties yet! | |||
Proven Targets:
| CHEMBL ID | UniProt ID | Name | Min activity | Assay type | Source |
|---|---|---|---|---|---|
| CHEMBL1293255 | P15428 | 15-hydroxyprostaglandin dehydrogenase [NAD+] |
31622.8 nM |
Potency |
via CMAUP
|
| CHEMBL3577 | P00352 | Aldehyde dehydrogenase 1A1 |
39810.7 nM 39810.7 nM |
Potency Potency |
via CMAUP
via CMAUP |
| CHEMBL218 | P21554 | Cannabinoid CB1 receptor |
3150 nM |
Ki |
PMID: 24900561
|
| CHEMBL253 | P34972 | Cannabinoid CB2 receptor |
1440 nM |
Ki |
PMID: 24900561
|
| CHEMBL1293226 | B2RXH2 | Lysine-specific demethylase 4D-like |
28183.8 nM |
Potency |
via CMAUP
|
| CHEMBL2608 | P10253 | Lysosomal alpha-glucosidase |
31622.8 nM |
Potency |
via CMAUP
|
| CHEMBL1293224 | P10636 | Microtubule-associated protein tau |
562.3 nM 22387.2 nM 28183.8 nM |
Potency Potency Potency |
via Super-PRED
via CMAUP via CMAUP |
| CHEMBL5162 | Q6W5P4 | Neuropeptide S receptor |
12589.3 nM |
Potency |
via CMAUP
|
Predicted Targets (via Super-PRED):
| CHEMBL ID | UniProt ID | Name | Probability | Model accuracy |
|---|---|---|---|---|
| CHEMBL1951 | P21397 | Monoamine oxidase A | 97.39% | 91.49% |
| CHEMBL5619 | P27695 | DNA-(apurinic or apyrimidinic site) lyase | 94.45% | 91.11% |
| CHEMBL3492 | P49721 | Proteasome Macropain subunit | 92.87% | 90.24% |
| CHEMBL2581 | P07339 | Cathepsin D | 91.50% | 98.95% |
| CHEMBL4208 | P20618 | Proteasome component C5 | 84.17% | 90.00% |
| CHEMBL3194 | P02766 | Transthyretin | 83.66% | 90.71% |
| CHEMBL3060 | Q9Y345 | Glycine transporter 2 | 82.12% | 99.17% |
| CHEMBL3251 | P19838 | Nuclear factor NF-kappa-B p105 subunit | 81.34% | 96.09% |
| CHEMBL3108638 | O15164 | Transcription intermediary factor 1-alpha | 80.91% | 95.56% |
| CHEMBL3401 | O75469 | Pregnane X receptor | 80.50% | 94.73% |
Below are displayed all the plants proven (via scientific papers) to contain this
compound!
To see more specific details click the taxa you are interested in.
To see more specific details click the taxa you are interested in.
| PubChem | 72300 |
| NPASS | NPC288411 |
| ChEMBL | CHEMBL180920 |
| LOTUS | LTS0194504 |
| wikiData | Q6732002 |