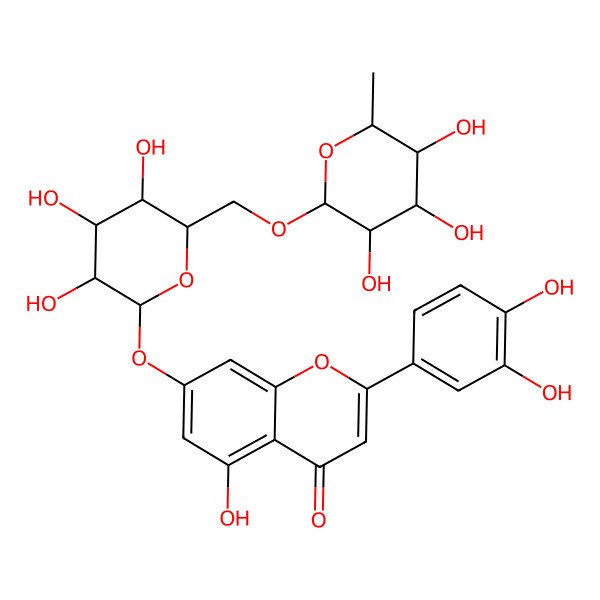
| 2-(3,4-dihydroxyphenyl)-5-hydroxy-7-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-[[(2R,3R,4R,5R,6S)-3,4,5-trihydroxy-6-methyloxan-2-yl]oxymethyl]oxan-2-yl]oxychromen-4-one |
| 2-(3,4-Dihydroxyphenyl)-5-hydroxy-7-((2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(((2R,3R,4R,5R,6S)-3,4,5-trihydroxy-6-methyloxan-2-yl)oxymethyl)oxan-2-yl)oxychromen-4-one |
| RefChem:154393 |
| 7-(((2S,3R,4S,5S,6R)-4,5-dihydroxy-6-(hydroxymethyl)-3-(((2S,3R,4R,5R,6S)-3,4,5-trihydroxy-6-methyloxan-2-yl)oxy)oxan-2-yl)oxy)-2-(3,4-dihydroxyphenyl)-4H-chromen-4-one |
| Scolymoside |
| 20633-84-5 |
| Luteolin-7-rutinoside |
| Luteolin 7-rutinoside |
| Luteolin 7-O-rutinoside |
| Scolimoside |
| Skolimoside |
| 7-Rutinosylluteolin |
| MLS002473221 |
| luteolin-7-O-beta-rutinoside |
| SMR001397309 |
| UNII-LDM9NL7QUS |
| orb1709233 |
| Luteolin-7-o-beta-D-rutinoside |
| CHEMBL1714943 |
| SCHEMBL23870123 |
| BDBM94850 |
| cid_10461109 |
| DTXSID201313731 |
| HMS2205K19 |
| EX-A6772 |
| HY-N6647 |
| AKOS040733634 |
| DA-75164 |
| FL158250 |
| LUTEOLIN-7-O-.BETA.-D-RUTINOSIDE |
| MS-30557 |
| CS-0066966 |
| G60943 |
| Luteolin 7-rutinoside, >=95% (LC/MS-ELSD) |
| SR-01000870981 |
| SR-01000870981-2 |
| Luteolin 7-o-alpha-L-rhamnopyranosyl-(1->6)-beta-D-glucopyranoside |
| LUTEOLIN 7-O-.ALPHA.-L-RHAMNOPYRANOSYL-(1->6)-.BETA.-D-GLUCOPYRANOSIDE |
| 2-(3,4-Dihydroxyphenyl)-5-hydroxy-7-(((2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-((((2R,3R,4R,5R,6S)-3,4,5-trihydroxy-6-methyltetrahydro-2H-pyran-2-yl)oxy)methyl)tetrahydro-2H-pyran-2-yl)oxy)-4H-chromen-4-one |
| 2-(3,4-dihydroxyphenyl)-5-hydroxy-7-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-[[(2R,3R,4R,5R,6S)-3,4,5-trihydroxy-6-methyl-tetrahydropyran-2-yl]oxymethyl]tetrahydropyran-2-yl]oxy-chromen-4-one |
| 2-(3,4-dihydroxyphenyl)-5-hydroxy-7-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-[[(2R,3R,4R,5R,6S)-3,4,5-trihydroxy-6-methyl-tetrahydropyran-2-yl]oxymethyl]tetrahydropyran-2-yl]oxy-chromone |
| 2-(3,4-dihydroxyphenyl)-5-hydroxy-7-[[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-[[(2R,3R,4R,5R,6S)-3,4,5-trihydroxy-6-methyl-2-oxanyl]oxymethyl]-2-oxanyl]oxy]-1-benzopyran-4-one |
| 2-[3,4-bis(oxidanyl)phenyl]-7-[(2S,3R,4S,5S,6R)-6-[[(2R,3R,4R,5R,6S)-6-methyl-3,4,5-tris(oxidanyl)oxan-2-yl]oxymethyl]-3,4,5-tris(oxidanyl)oxan-2-yl]oxy-5-oxidanyl-chromen-4-one |
| 4H-1-BENZOPYRAN-4-ONE, 7-((6-O-(6-DEOXY-.ALPHA.-L-MANNOPYRANOSYL)-.BETA.-D-GLUCOPYRANOSYL)OXY)-2-(3,4-DIHYDROXYPHENYL)-5-HYDROXY- |
| 4H-1-Benzopyran-4-one, 7-((6-O-(6-deoxy-alpha-L-mannopyranosyl)-beta-D-glucopyranosyl)oxy)-2-(3,4-dihydroxyphenyl)-5-hydroxy- |
|
There are more than 10 synonyms. If you wish to see them all click here.
|