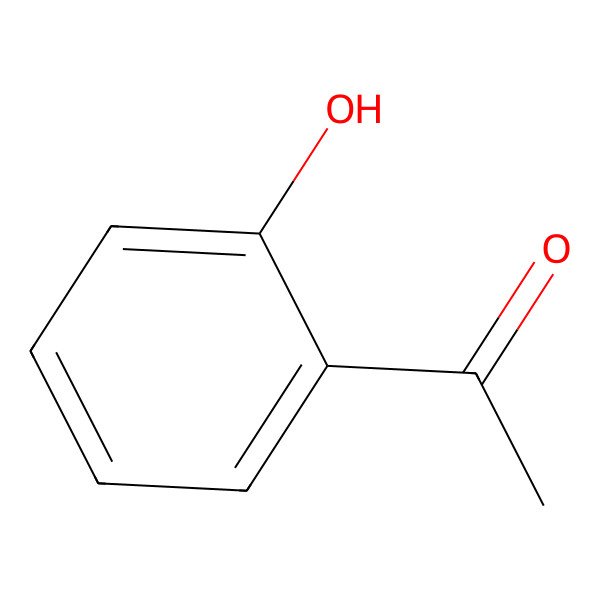
| 118-93-4 |
| 1-(2-Hydroxyphenyl)ethanone |
| 2-Acetylphenol |
| O-HYDROXYACETOPHENONE |
| o-Hydroxyphenyl methyl ketone |
| 1-(2-Hydroxyphenyl)Ethan-1-One |
| 2-Hydroxyphenyl methyl ketone |
| Ethanone, 1-(2-hydroxyphenyl)- |
| Methyl 2-hydroxyphenyl ketone |
| FEMA No. 3548 |
| DTXSID7040285 |
| NSC-16933 |
| 3E533Z76W0 |
| DTXCID5020285 |
| CHEBI:145716 |
| RefChem:908432 |
| 204-288-0 |
| 2-hydroxy-1-phenyl-ethanone |
| 2-hydroxy-1-phenylethanone |
| 2-Hydroxyacetophenone |
| Benzoylcarbinol |
| CHEBI:28341 |
| Glycolophenone |
| NSC-171232 |
| Phenacyl alcohol |
| o-Acetylphenol |
| 104809-67-8 |
| 2-Acetophenol |
| Acetophenone, 2'-hydroxy- |
| Acetophenone, o-hydroxy- |
| USAF KE-20 |
| 2/'-Hydroxyacetophenone |
| 125507-95-1 |
| MFCD00002219 |
| NSC 16933 |
| 2-Hydroxyacetylbenzene |
| FEMA 3548 |
| 1-(2-hydroxyphenyl)-ethanone |
| CHEMBL3187012 |
| 2,4-Cyclohexadien-1-one, 6-(1-hydroxyethylidene)- (9CI) |
| Ethanone,1-(2-hydroxyphenyl)-, labeled with carbon-14 (9CI) |
| 1-acetyl-2-hydroxybenzene |
| WLN: QR BV1 |
| Paracetamol EP Impurity I |
| Ethanone, 1-(hydroxyphenyl)- |
| 1189865-36-8 |
| O-ACETOPHENOL |
| 2'-hydroxy-acetophenone |
| EINECS 204-288-0 |
| BRN 0386123 |
| o-acetyl phenol |
| AI3-12134 |
| UNII-3E533Z76W0 |
| 1-(2-Hydroxyphenyl)ethanone; Paracetamol Imp. I (EP); 2-Hydroxyacetophenone; Paracetamol Impurity I |
| 2,4-Cyclohexadien-1-one, 6-(1-hydroxyethylidene)- |
| orthohydroxyacetophenone |
| Paracetamol Impurity I |
| 2`-Hydroxyacetophenone |
| 2\'-Hydroxyacetophenone |
| 1-(hydroxyphenyl)ethanone |
| 1-(2-Hydroxyphenyl)etanone |
| SCHEMBL40865 |
| 2-Acetophenol;2-Acetylphenol |
| 4-08-00-00320 (Beilstein Handbook Reference) |
| ACETOPHENONE,2-HYDROXY |
| SCHEMBL126876 |
| 1-(2-hydroxy-phenyl)ethanone |
| orb1297719 |
| SCHEMBL1893665 |
| SCHEMBL2266654 |
| SCHEMBL6326192 |
| 1-(2-hydroxy phenyl)-ethanone |
| 1-(2-hydroxy-phenyl)-ethanone |
| 2'-Hydroxyacetophenone - 95% |
| SCHEMBL30293783 |
| HY-Y1426R |
| TIMTEC-BB SBB040261 |
| NSC9263 |
| 2'-Hydroxyacetophenone, >=98% |
| OTAVA-BB 1778867 |
| 2'-Hydroxyacetophenone (Standard) |
| AKOS 90575 |
| LABOTEST-BB LTBB003445 |
| 1-(2-Hydroxyphenyl)ethanone, 9CI |
| Acetophenone, 2'-hydroxy- (8CI) |
| HY-Y1426 |
| KS-000002MM |
| NSC-9263 |
| NSC16933 |
| NSC44452 |
| PXB86536 |
| STR00372 |
| AKOS BBS-00003239 |
| Tox21_301119 |
| 2-HYDROXYACETOPHENONE [FHFI] |
| BBL012126 |
| BDBM50140214 |
| LABOTEST-BB LT03333567 |
| NSC-44452 |
| s9375 |
| SBB040261 |
| STL163508 |
| 2`-Hydroxyacetophenone 2-Acetylphenol |
| AKOS000118832 |
| AC-5622 |
| AS01159 |
| CCG-266129 |
| CS-W020051 |
| FH64535 |
| PS-3396 |
| TRA0030570 |
| NCGC00248294-01 |
| NCGC00257527-01 |
| AK-44482 |
| BP-13220 |
| CAS-118-93-4 |
| FH171307 |
| 2'-Hydroxyacetophenone, analytical standard |
| DB-269423 |
| AM20060222 |
| H0192 |
| NS00020731 |
| PARACETAMOL IMPURITY I [EP IMPURITY] |
| ST50213425 |
| EN300-18385 |
| 2'-Hydroxyacetophenone, ReagentPlus(R), 99% |
| D70588 |
| M-7235 |
| 2'-Hydroxyacetophenone (Acetaminophen Impurity I) |
| F078584 |
| doi:10.14272/JECYUBVRTQDVAT-UHFFFAOYSA-N.1 |
| Q27257104 |
| Z57101014 |
| 2'-Hydroxyacetophenone, Vetec(TM) reagent grade, 98% |
| F1908-0162 |
| InChI=1/C8H8O2/c1-6(9)7-4-2-3-5-8(7)10/h2-5,10H,1H |
|
There are more than 10 synonyms. If you wish to see them all click here.
|