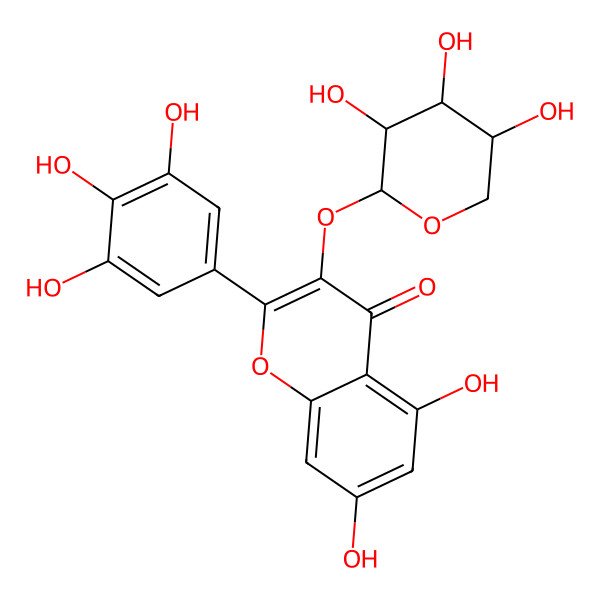
Myricetin-3-O-xyloside
| Internal ID | 1a80b007-5dba-4c6a-9c49-82e91c165d7d |
| Taxonomy | Phenylpropanoids and polyketides > Flavonoids > Flavonoid glycosides > Flavonoid O-glycosides > Flavonoid-3-O-glycosides |
| IUPAC Name | 5,7-dihydroxy-3-(3,4,5-trihydroxyoxan-2-yl)oxy-2-(3,4,5-trihydroxyphenyl)chromen-4-one |
| SMILES (Canonical) | C1C(C(C(C(O1)OC2=C(OC3=CC(=CC(=C3C2=O)O)O)C4=CC(=C(C(=C4)O)O)O)O)O)O |
| SMILES (Isomeric) | C1C(C(C(C(O1)OC2=C(OC3=CC(=CC(=C3C2=O)O)O)C4=CC(=C(C(=C4)O)O)O)O)O)O |
| InChI | InChI=1S/C20H18O12/c21-7-3-8(22)13-12(4-7)31-18(6-1-9(23)14(26)10(24)2-6)19(16(13)28)32-20-17(29)15(27)11(25)5-30-20/h1-4,11,15,17,20-27,29H,5H2 |
| InChI Key | SBEOEJNITMVWLK-UHFFFAOYSA-N |
| Popularity | 5 references in papers |
| Molecular Formula | C20H18O12 |
| Molecular Weight | 450.30 g/mol |
| Exact Mass | 450.07982601 g/mol |
| Topological Polar Surface Area (TPSA) | 207.00 Ų |
| XlogP | 0.10 |
| CHEBI:192520 |
| Hexahydroxy-flavone (myricetin) pentoside |
| 5,7-dihydroxy-3-(3,4,5-trihydroxyoxan-2-yl)oxy-2-(3,4,5-trihydroxyphenyl)chromen-4-one |
| Target | Value | Probability (raw) | Probability (%) |
|---|---|---|---|
| No predicted properties yet! | |||
Proven Targets:
| CHEMBL ID | UniProt ID | Name | Min activity | Assay type | Source |
|---|---|---|---|---|---|
| No proven targets yet! | |||||
Predicted Targets (via Super-PRED):
| CHEMBL ID | UniProt ID | Name | Probability | Model accuracy |
|---|---|---|---|---|
| CHEMBL5619 | P27695 | DNA-(apurinic or apyrimidinic site) lyase | 99.76% | 91.11% |
| CHEMBL1806 | P11388 | DNA topoisomerase II alpha | 97.09% | 89.00% |
| CHEMBL2345 | P51812 | Ribosomal protein S6 kinase alpha 3 | 94.67% | 95.64% |
| CHEMBL2635 | P51452 | Dual specificity protein phosphatase 3 | 93.33% | 94.00% |
| CHEMBL1951 | P21397 | Monoamine oxidase A | 92.09% | 91.49% |
| CHEMBL2581 | P07339 | Cathepsin D | 89.61% | 98.95% |
| CHEMBL3194 | P02766 | Transthyretin | 89.25% | 90.71% |
| CHEMBL5845 | P23415 | Glycine receptor subunit alpha-1 | 88.50% | 90.71% |
| CHEMBL1860 | P10827 | Thyroid hormone receptor alpha | 88.18% | 99.15% |
| CHEMBL3108638 | O15164 | Transcription intermediary factor 1-alpha | 87.37% | 95.56% |
| CHEMBL3137262 | O60341 | LSD1/CoREST complex | 86.19% | 97.09% |
| CHEMBL1075162 | Q13304 | Uracil nucleotide/cysteinyl leukotriene receptor | 85.01% | 80.33% |
| CHEMBL3038477 | P67870 | Casein kinase II alpha/beta | 84.02% | 99.23% |
| CHEMBL241 | Q14432 | Phosphodiesterase 3A | 83.04% | 92.94% |
| CHEMBL3060 | Q9Y345 | Glycine transporter 2 | 83.03% | 99.17% |
| CHEMBL4203 | Q9HAZ1 | Dual specificity protein kinase CLK4 | 82.48% | 94.45% |
| CHEMBL1293249 | Q13887 | Kruppel-like factor 5 | 82.04% | 86.33% |
| CHEMBL5339 | Q5NUL3 | G-protein coupled receptor 120 | 81.80% | 95.78% |
| CHEMBL3401 | O75469 | Pregnane X receptor | 81.66% | 94.73% |
Below are displayed all the plants proven (via scientific papers) to contain this
compound!
To see more specific details click the taxa you are interested in.
To see more specific details click the taxa you are interested in.
| Epilobium hirsutum |
| Eucalyptus camaldulensis subsp. camaldulensis |
| Limonium aureum |
| Limonium gmelinii |
| Limonium sinense |
| Myrsine africana |
| Saxifraga tricuspidata |
| Vaccinium macrocarpon |
| PubChem | 21477996 |
| LOTUS | LTS0072077 |
| wikiData | Q105249363 |