| 41753-43-9 |
| Gypenoside III |
| Gynosaponin C |
| Sanchinoside E1 |
| Arasaponin E1 |
| Panax saponin E |
| Pseudoginsenoside D |
| ginsenoside-Rb1 |
| Panaxsaponin E |
| Panaxoside Rb1 |
| Sanchinoside Rb1 |
| Notoginsenoside Rb1 |
| 7413S0WMH6 |
| GRb 1 |
| CHEBI:67989 |
| DTXSID401316929 |
| NSC-310103 |
| 3-GlcGlc-20-GlcGlc-ginsenoside |
| RefChem:37701 |
| DTXCID601746750 |
| 255-532-8 |
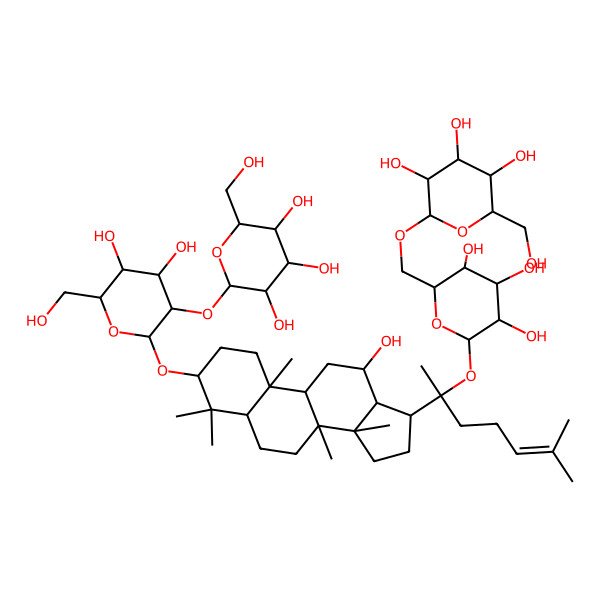
| (2R,3R,4S,5S,6R)-2-(((2R,3S,4S,5R,6S)-6-((1S)-1-((3S,5R,8R,9R,10R,12R,13R,14R,17S)-3-((2R,3R,4S,5S,6R)-4,5-dihydroxy-6-(hydroxymethyl)-3-((2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)tetrahydropyran-2-yl)oxy-tetrahydropyran-2-yl)oxy-12-hydroxy-4,4,8,10,14-pentamethyl-2,3,5,6,7,9,11,12,13,15,16,17-dodecahydro-1H-cyclopenta(a)phenanthren-17-yl)-1,5-dimethyl-hex-4-enoxy)-3,4,5-trihydroxy-tetrahydropyran-2-yl)methoxy)-6-(hydroxymethyl)tetrahydropyran-3,4,5-triol |
| (2R,3R,4S,5S,6R)-2-(((2R,3S,4S,5R,6S)-6-((2S)-2-((3S,5R,8R,9R,10R,12R,13R,14R,17S)-3-((2R,3R,4S,5S,6R)-4,5-dihydroxy-6-(hydroxymethyl)-3-((2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl)oxyoxan-2-yl)oxy-12-hydroxy-4,4,8,10,14-pentamethyl-2,3,5,6,7,9,11,12,13,15,16,17-dodecahydro-1H-cyclopenta(a)phenanthren-17-yl)-6-methylhept-5-en-2-yl)oxy-3,4,5-trihydroxyoxan-2-yl)methoxy)-6-(hydroxymethyl)oxane-3,4,5-triol |
| MFCD00133367 |
| (20S)-ginsenoside Rb1 |
| CHEMBL501515 |
| 2-O-beta-Glucopyranosyl-(3beta,12beta)-20-[(6-O-beta-D-glucopyranosyl-beta-D-glucopyranosyl)oxy]-12-hydroxydammar-24-en-3-yl beta-D-glucopyranoside |
| GinsenosideRb1 |
| EINECS 255-532-8 |
| 2-O-beta-Glucopyranosyl-(3beta,12beta)-20-((6-O-beta-D-glucopyranosyl-beta-D-glucopyranosyl)oxy)-12-hydroxydammar-24-en-3-yl-beta-D-glucopyranoside |
| GS-Rb1 |
| C54H92O23 |
| UNII-7413S0WMH6 |
| NSC 310103 |
| GSRb1 |
| 20(S)-ginsenoside Rb1 |
| Ginsenoside Rb1 - 94% |
| Ginsenoside Rb1 - 98% |
| Ginsenoside Rb1 (Standard) |
| BIDD:ER0108 |
| orb1304975 |
| SCHEMBL29350543 |
| HY-N0039R |
| GINSENOSIDE RB1 [USP-RS] |
| GINSENOSIDE RB1 [WHO-DD] |
| GLXC-19161 |
| HMS3885O12 |
| HMS6018L20 |
| EX-A6786 |
| HY-N0039 |
| MSK40041 |
| BDBM50317541 |
| s3924 |
| AKOS025311537 |
| CCG-270640 |
| CS-3829 |
| DB06749 |
| OG09288 |
| NCGC00347398-02 |
| NCGC00347398-04 |
| BS-32417 |
| LS-71528 |
| SY068870 |
| XG164977 |
| 13 - Ginseng supplement chemical analysis |
| C20713 |
| 753G439 |
| Ginsenoside Rb1, primary pharmaceutical reference standard |
| Ginsenoside Rb1, European Pharmacopoeia (EP) Reference Standard |
| (3b,12b)-20-[(6-O-b-D-Glucopyranosyl-b-D-glucopyranosyl)oxy]-12-hydroxydammar-24-en-3-yl 2-O-(b-D-glucopyranosyl)-b-D-glucopyranosid e;Arasaponin E1;Gypenoside III |
| (3beta,12beta)-20-[(6-O-beta-D-glucopyranosyl-beta-D-glucopyranosyl)oxy]-12-hydroxydammar-24-en-3-yl 2-O-beta-D-glucopyranosyl-beta-D-glucopyranoside |
| (3beta,12beta)-20-{[6-O-(beta-D-glucopyranosyl)-beta-D-glucopyranosyl]oxy}-12-hydroxydammar-24-en-3-yl 2-O-beta-D-glucopyranosyl-beta-D-glucopyranoside |
| .BETA.-D-GLUCOPYRANOSIDE, (3.BETA.,12.BETA.)-20-((6-O-.BETA.-D-GLUCOPYRANOSYL-.BETA.-D-GLUCOPYRANOSYL)OXY)-12-HYDROXYDAMMAR-24-EN-3-YL 2-O-.BETA.-D-GLUCOPYRANOSYL- |
| 3beta-[beta-D-glucopyranosyl-(1->2)-beta-D glucopyranosyloxy]-20-[beta-D-glucopyranosyl-(1->2)-beta-D glucopyranosyloxy]dammar-24-en-12beta-ol |
| Arasaponin E1;Gypenoside III;(3b,12b)-20-[(6-O-b-D-Glucopyranosyl-b-D-glucopyranosyl)oxy]-12-hydroxydammar-24-en-3-yl 2-O-(b-D-gluco pyranosyl)-b-D-glucopyranoside |
| beta-D-Glucopyranoside, (3-beta,12-beta)-20-((6-O-beta-D-glucopyranosyl-beta-D-glucopyranosyl)oxy)-12-hydroxydammar-24-en-3-yl 2-O-beta-D-glucopyranosyl- |
| GINSENOSIDE RB1 (CONSTITUENT OF AMERICAN GINSENG, ASIAN GINSENG, AND TIENCHI GINSENG) [DSC] |
|
There are more than 10 synonyms. If you wish to see them all click here.
|