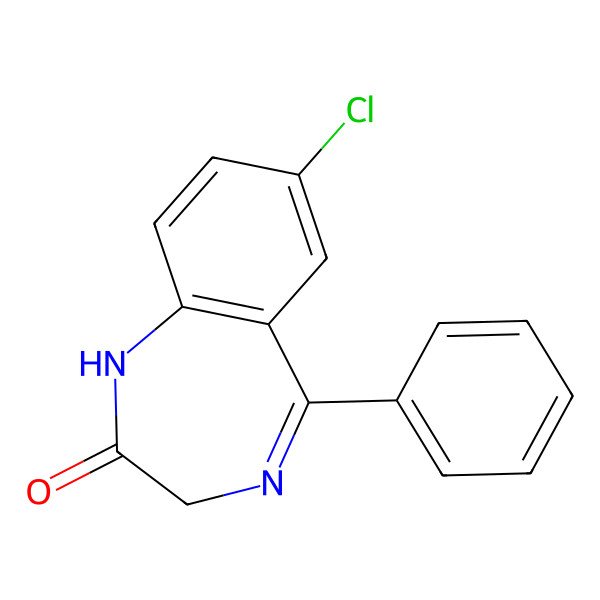
| Nordiazepam |
| desmethyldiazepam |
| Norprazepam |
| N-Desmethyldiazepam |
| 1088-11-5 |
| Calmday |
| N-Demethyldiazepam |
| N-Deoxydemoxepam |
| Madar |
| 1-Demethyldiazepam |
| N1-Desmethyldiazepam |
| Demethyldiazepam |
| Demadar |
| Praxadium |
| Stilny |
| Nordazepamum |
| Ro 5-2180 |
| DMDZ |
| N-Deoxydemoxapam |
| Nordazepam CIV |
| Nordazepam [INN] |
| 7-Chloro-1,3-dihydro-5-phenyl-2H-1,4-benzodiazepin-2-one |
| N-Descyclopropylmethylprazepam |
| 7-chloro-5-phenyl-1,3-dihydro-1,4-benzodiazepin-2-one |
| A 101 |
| A101 |
| 2H-1,4-Benzodiazepin-2-one, 7-chloro-1,3-dihydro-5-phenyl- |
| NSC 46078 |
| 7-Chloro-5-phenyl-1,3-dihydro-2H-1,4-benzodiazepin-2-one |
| NSC-46078 |
| CHEMBL523 |
| NSC-631619 |
| NDZ |
| DTXSID2049000 |
| Vegesan |
| 2H-1,4-Benzodiazepin-2-one, 1,3-dihydro-7-chloro-5-phenyl- |
| CHEBI:111762 |
| Nordazepam (INN) |
| NSC631619 |
| 67220MCM01 |
| NDD |
| NCGC00168263-01 |
| Nordaz |
| 7-Chloro-1,3-dihydro-5-phenyl-(2H)-1,4-benzodiazepin-2-one |
| 7-Chloro-5-phenyl-1,3-dihydro-benzo[e][1,4]diazepin-2-one |
| Lomax |
| Sopax |
| Desmethyldiazepam (Nordazepam) |
| Nordazepamum [INN-Latin] |
| CCRIS 9181 |
| EINECS 214-123-4 |
| BRN 0751823 |
| Desoxydemoxepam |
| DEA No. 2838 |
| UNII-67220MCM01 |
| Calmday (TN) |
| Ro 52180 |
| 7-Chlor-2,3-dihydro-5-phenyl-1H-1,4-benzodiazepin-2-on |
| Diazepam-M N-desmethyl |
| Diazepam EP Impurity A |
| NORDAZEPAM [MI] |
| ChemDiv1_028362 |
| NORDAZEPAM [MART.] |
| MixCom6_000554 |
| NORDAZEPAM [WHO-DD] |
| Oprea1_578139 |
| SCHEMBL78453 |
| 5-24-04-00291 (Beilstein Handbook Reference) |
| MLS001173623 |
| DivK1c_000979 |
| DTXCID3028926 |
| NORDAZEPAM CIV [USP-RS] |
| HMS503C19 |
| HMS667J04 |
| KBio1_000979 |
| NINDS_000979 |
| HMS2272B05 |
| NSC46078 |
| Tox21_113493 |
| BDBM50027835 |
| Desmethyldiazepam, analytical standard |
| WLN: T67 GMV JN IHJ CG KR |
| AKOS000636746 |
| AKOS005486646 |
| DB14028 |
| IDI1_000979 |
| DIAZEPAM IMPURITY A [EP IMPURITY] |
| SMR000238153 |
| CAS-1088-11-5 |
| 2H-1, 1,3-dihydro-7-chloro-5-phenyl- |
| 2H-1, 7-chloro-1,3-dihydro-5-phenyl- |
| 5-phenyl-7-chloro-1,4-benzodiazepin-2-one |
| C07486 |
| D08283 |
| 7-Chloro-5-phenyl-3H-1,4-benzodiazepin-2-one |
| Nordazepam (Nordiazepam) 0.1 mg/ml in Methanol |
| Nordazepam (Nordiazepam) 1.0 mg/ml in Methanol |
| A801929 |
| Desmethyldiazepam (Nordazepam), 1mg/ml in Methanol |
| Q3180288 |
| DIPOTASSIUM CLORAZEPATE IMPURITY B [EP IMPURITY] |
| 7-chloro-5-phenyl-1,3-dihydro-[1,4]benzodiazepin-2-one |
| 7-chloro-5-phenyl-1H-benzo[e][1,4]diazepin-2(3H)-one |
| (z)-7-chloro-5-phenyl-1h-benzo[e][1,4]diazepin-2(3h)-one |
| 7-chloro-1,3 -dihydro-5-phenyl-2H-1,4-benzodiazepine-2-one |
| 7-chloro-2,3-dihydro-5-phenyl-1H-1,4-benzodiazepin-2-one |
| 7-Chloro-5-phenyl-1,3-dihydro-2H-1,4-benzodiazepin-2-one # |
| 7-Chloro-5-phenyl-1,3-dihydro-benzo[e][1,4]diazepin-2-one( am) |
| 7-Chloro-5-phenyl-2-oxo-2,3-dihydro-1H-benzo[f]-1,4-diazepine |
| 7-Chloro-5-phenyl-1,3-dihydro-2H-1,4-benzodiazepin-2-one (Nordazepam) |
| 7-Chloro-5-phenyl-1,3-dihydro-benzo[e][1,4]diazepin-2-one(nordiazepam) |
| 7-Chloro-5-phenyl-1,3-dihydro-benzo[e][1,4]diazepin-2-one(desmethyldiazepam) |
| Nordazepam; DesMethyl Amino Diazepam; 7-chloro-5-phenyl-1,3-dihydro-2H-1,4-benzodiazepin-2-one |
|
There are more than 10 synonyms. If you wish to see them all click here.
|