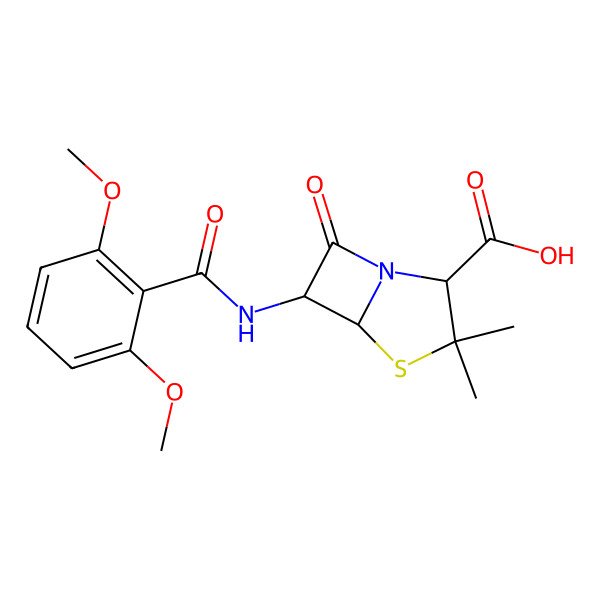
| Meticillin |
| Methycillin |
| 61-32-5 |
| Methicillinum |
| Meticilina |
| Meticilline |
| Meticillinum |
| Staphcillin |
| Metacillin |
| Dimocillin |
| Meticilina [INN-Spanish] |
| Meticilline [INN-French] |
| Meticillinum [INN-Latin] |
| (2,6-Dimethoxyphenyl)penicillin |
| 6-(2,6-Dimethoxybenzamido)penicillanic acid |
| CHEBI:6827 |
| (2S,5R,6R)-6-[(2,6-dimethoxybenzoyl)amino]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid |
| BRL 1241 |
| 6beta-(2,6-dimethoxybenzamido)penicillanic acid |
| 2,6-dimethoxyphenyl penicillin |
| Meticillina |
| Q91FH1328A |
| Celbenin |
| (2S,5R,6R)-6-(2,6-dimethoxybenzamido)-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid |
| Meticillina [DCIT] |
| Methcillin |
| Meticillin [INN] |
| Methicillin [USAN] |
| HSDB 3121 |
| Meticillin [INN:BAN] |
| EINECS 200-505-8 |
| Penicillin, (2,6-dimethoxyphenyl)- |
| UNII-Q91FH1328A |
| (2S,5R,6R)-6-(2,6-DIMETHOXYBENZAMIDO)-3,3-DIMETHYL-7-OXO-4-THIA-1-AZABICYCLO(3.2.0)HEPTANE-2-CARBOXYLIC ACID |
| 4-Thia-1-azabicyclo(3.2.0)heptane-2-carboxylic acid, 6-((2,6-dimethoxybenzoyl)amino)-3,3-dimethyl-7-oxo-, (2S-(2alpha,5alpha,6beta))- |
| Spectrum_000993 |
| Spectrum2_001965 |
| Spectrum3_000494 |
| Spectrum4_000878 |
| Spectrum5_001600 |
| METICILLIN [HSDB] |
| CHEMBL575 |
| Epitope ID:139649 |
| METHICILLIN [VANDF] |
| SCHEMBL4898 |
| METICILLIN [WHO-DD] |
| BSPBio_001987 |
| KBioGR_001575 |
| KBioSS_001473 |
| 4-Thia-1-azabicyclo(3.2.0)heptane-2-carboxylic acid, 6-(2,6-dimethoxybenzamido)-3,3-dimethyl-7-oxo- |
| 6-(2,3-Dimethoxybenzamido)-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo(3.2.0)heptane-2-carboxylic acid |
| DivK1c_000100 |
| SPBio_002089 |
| DTXSID6023284 |
| GTPL12264 |
| KBio1_000100 |
| KBio2_001473 |
| KBio2_004041 |
| KBio2_006609 |
| KBio3_001487 |
| NINDS_000100 |
| RJQXTJLFIWVMTO-TYNCELHUSA-N |
| BDBM50103523 |
| 6beta-(2,6-dimethoxybenzamido)-2,2-dimethylpenam-3alpha-carboxylic acid |
| AKOS030530774 |
| DB01603 |
| IDI1_000100 |
| (2S,5R,6R)-6-{[(2,6-dimethoxyphenyl)carbonyl]amino}-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid |
| 4-Thia-1-azabicyclo(3.2.0)heptane-2-carboxylic acid, 6-(2,6-dimethoxybenzamido)-3,3,-dimethyl-7-oxo- |
| MII |
| SBI-0051440.P003 |
| HY-121544 |
| MRSA Selective Supplement, for microbiology |
| CS-0082724 |
| C07177 |
| EN300-19748934 |
| Q409262 |
| BRD-K34388247-236-02-5 |
| (2S,5R,6R)-6-[(2,6-dimethoxybenzene)amido]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid |
|
There are more than 10 synonyms. If you wish to see them all click here.
|