| Excolabdone C |
| Coleonol B |
| 64657-21-2 |
| UNII-256AFQ3YUN |
| 256AFQ3YUN |
| Isoforskolin (constituent of forskohlii) [DSC] |
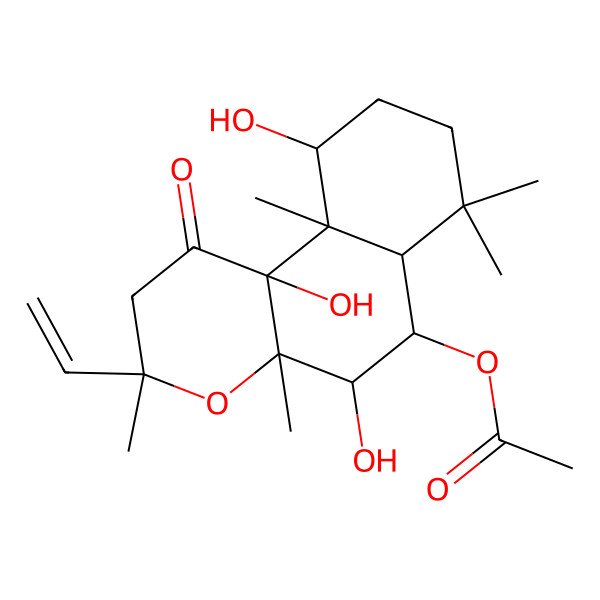
| 6beta-acetoxy-8,13-epoxy-1alpha,7beta,9alpha-trihydroxylabd-14-en-11-one |
| [(3R,4aR,5S,6S,6aS,10S,10aR,10bS)-3-ethenyl-5,10,10b-trihydroxy-3,4a,7,7,10a-pentamethyl-1-oxo-5,6,6a,8,9,10-hexahydro-2H-benzo[f]chromen-6-yl] acetate |
| 1H-Naphtho(2,1-b)pyran-1-one, 6-(acetyloxy)-3-ethenyldodecahydro-5,10,10b-trihydroxy-3,4a,7,7,10a-pentamethyl-, (3R-(3alpha,4abeta,5beta,6beta,6aalpha,10alpha,10abeta,10balpha))- |
| 6.BETA.-ACETOXY-8,13-EPOXY-1.ALPHA.,7.BETA.,9.ALPHA.-TRIHYDROXYLABD-14-EN-11-ONE |
| 1H-NAPHTHO(2,1-B)PYRAN-1-ONE, 6-(ACETYLOXY)-3-ETHENYLDODECAHYDRO-5,10,10B-TRIHYDROXY-3,4A,7,7,10A-PENTAMETHYL-, (3R-(3.ALPHA.,4A.BETA.,5.BETA.,6.BETA.,6A.ALPHA.,10.ALPHA.,10A.BETA.,10B.ALPHA.))- |
| ((3R,4aR,5S,6S,6aS,10S,10aR,10bS)-3-ethenyl-5,10,10b-trihydroxy-3,4a,7,7,10a-pentamethyl-1-oxo-5,6,6a,8,9,10-hexahydro-2H-benzo(f)chromen-6-yl) acetate |
| RefChem:149342 |
| ISOFORSKOLIN (CONSTITUENT OF FORSKOHLII) |
| Iso-Forskolin |
| MLS000517267 |
| SMR000127420 |
| (3R,4aR,5S,6S,6aS,10S,10aR,10bS)-5,10,10b-Trihydroxy-3,4a,7,7,10a-pentamethyl-1-oxo-3-vinyldodecahydro-1H-benzo[f]chromen-6-yl acetate |
| (3R,4aR,5S,6S,6aS,10S,10aR,10bS)-6-(Acetyloxy)-3-ethenyldodecahydro-5,10,10b-trihydroxy-3,4a,7,7,10a-pentamethyl-1H-naphtho[2,1-b]pyran-1-one |
| 6BETA-ACETOXY-8,13-EPOXY-1ALPHA,7BETA,9ALPHA-TRIHYDROXY-LABD-14-EN-11-ONE |
| MFCD00078929 |
| Isoforskolin (Standard) |
| orb1297414 |
| orb3139255 |
| SCHEMBL4928148 |
| CHEMBL1519619 |
| BDBM83744 |
| cid_9549169 |
| HY-N6927R |
| REGID_for_CID_9549169 |
| HMS2267A05 |
| HY-N6927 |
| TN1520 |
| AKOS037514904 |
| EBC-615437 |
| (3R,4aR,5S,6S,6aS,10S,10aR,10bS)-3-ethenyl-5,10,10b-trihydroxy-3,4a,7,7,10a-pentamethyl-1-oxo-dodecahydro-1H-naphtho[2,1-b]pyran-6-yl acetate |
| AC-35072 |
| MS-27083 |
| CS-0027682 |
| Q27253921 |
| [(3R,4aR,5S,6S,6aS,10S,10aR,10bS)-3-ethenyl-3,4a,7,7,10a-pentamethyl-5,10,10b-tris(oxidanyl)-1-oxidanylidene-5,6,6a,8,9,10-hexahydro-2H-benzo[f]chromen-6-yl] ethanoate |
| [(3R,4aR,5S,6S,6aS,10S,10aR,10bS)-5,10,10b-trihydroxy-3,4a,7,7,10a-pentamethyl-1-oxo-3-vinyl-5,6,6a,8,9,10-hexahydro-2H-benzo[f]chromen-6-yl] acetate |
| acetic acid [(3R,4aR,5S,6S,6aS,10S,10aR,10bS)-3-ethenyl-5,10,10b-trihydroxy-3,4a,7,7,10a-pentamethyl-1-oxo-5,6,6a,8,9,10-hexahydro-2H-benzo[f][1]benzopyran-6-yl] ester |
| acetic acid [(3R,4aR,5S,6S,6aS,10S,10aR,10bS)-5,10,10b-trihydroxy-1-keto-3,4a,7,7,10a-pentamethyl-3-vinyl-5,6,6a,8,9,10-hexahydro-2H-benzo[f]chromen-6-yl] ester |
|
There are more than 10 synonyms. If you wish to see them all click here.
|