| 497-30-3 |
| L-Ergothioneine |
| Sympectothion |
| Ergothionine |
| Thioneine |
| l-Thioneine |
| Thiasine |
| L(+)-Ergothioneine |
| Thiolhistidine-betaine |
| BDZ3DQM98W |
| DTXSID901020082 |
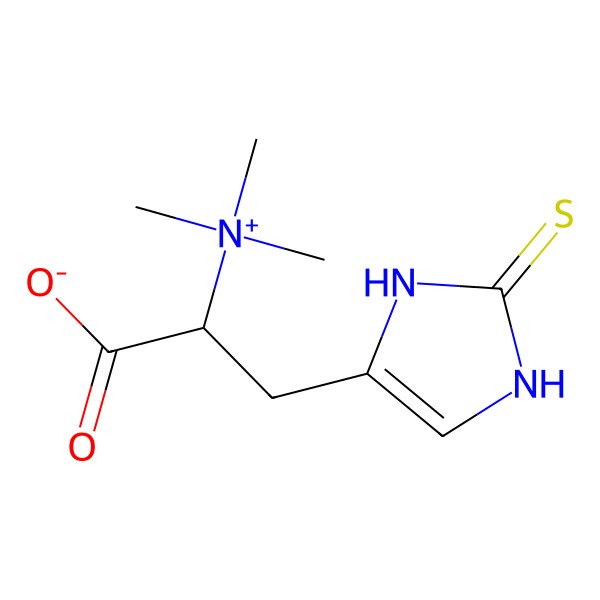
| (2S)-3-(2-sulfanylidene-1,3-dihydroimidazol-4-yl)-2-(trimethylazaniumyl)propanoate |
| CHEBI:4828 |
| (2S)-3-(2-sulfanylidene-2,3-dihydro-1H-imidazol-4-yl)-2-(trimethylazaniumyl)propanoate |
| 1H-Imidazole-4-ethanaminium, alpha-carboxy-2,3-dihydro-N,N,N-trimethyl-2-thioxo-, inner salt, (S)- |
| 2 Thiol L histidine betaine |
| (alphaS)-alpha-carboxy-2,3-dihydro-N,N,N-trimethyl-2-thioxo-1H-imidazole-4-ethanaminium inner salt |
| NSC-7175 |
| 2-Thiol-L-histidine-betaine |
| RefChem:591076 |
| DTXCID901477922 |
| 5-((2S)-2-carboxy-2-(trimethylazaniumyl)ethyl)-1H-imidazole-2-thiolate |
| 207-843-5 |
| L-(+)-Ergothioneine |
| (S)-3-(2-Thioxo-2,3-dihydro-1H-imidazol-4-yl)-2-(trimethylammonio)propanoate |
| NSC 7175 |
| MFCD00167474 |
| ergothioneine (thione form) |
| Thioeine |
| (S)-(1-Carboxy-2-(2-mercaptoimidazol-4-yl)ethyl)trimethylammonium hydroxide |
| NSC7175 |
| UNII-BDZ3DQM98W |
| erythrothioneine |
| thiolhistidinebetaine |
| EINECS 207-843-5 |
| ERGOLD |
| Ergothioneine L-(+) |
| ergothioneine thione form |
| AI3-23492 |
| ERGOTHIONEINE [MI] |
| 2-Mercaptohistidine Betaine |
| SCHEMBL188140 |
| SCHEMBL188141 |
| orb1299136 |
| orb3012990 |
| SCHEMBL9985141 |
| CHEMBL5209630 |
| CHEBI:82707 |
| HMS2089D04 |
| 2-mercaptohistidine trimethylbetaine |
| HY-N1914 |
| MSK169210 |
| AKOS027325505 |
| CCG-207908 |
| EBC-616355 |
| FE22785 |
| 1H-Imidazole-4-ethanaminium, .alpha.-carboxy-2,3-dihydro-N,N,N-trimethyl-2-thioxo-, inner salt, (.alpha.S)- |
| 1H-Imidazole-4-ethanaminium, alpha-carboxy-2,3-dihydro-N,N,N-trimethyl-2-thioxo-, inner salt, (alphaS)- |
| DA-64818 |
| SY382275 |
| CS-0018211 |
| D83035 |
| AB01275464-01 |
| Ergothionine;L-(+)-Ergothioneine;Erythrothioneine |
| Q614788 |
| (2S)-3-(2-mercapto-1H-imidazol-5-yl)-2-(trimethylammonio)propanoate |
| (2S)-3-(2-mercapto-1H-imidazol-5-yl)-2-(trimethylazaniumyl)propanoate |
| (2S)-3-(2-sulfanyl-1H-imidazol-4-yl)-2-(trimethylazaniumyl)propanoate |
| 3-(2-sulfanylidene-1,3-dihydroimidazol-4-yl)-2-trimethylammonio-propanoate |
| Nalpha,Nalpha,Nalpha-trimethyl-2-sulfanylidene-2,3-dihydro-L-histidine |
| (2S)-3-(2-Thioxo-2,3-dihydro-1H-imidazol-4-yl)-2-(trimethylammonio)propanoate |
| (2S)-3-(2-thioxo-2,3-dihydro-1H-imidazol-4-yl)-2-(trimethylazaniumyl)propanoate |
| (.ALPHA.S)-.ALPHA.-CARBOXY-2,3-DIHYDRO-N,N,N-TRIMETHYL-2-THIOXO-1H-IMIDAZOLE-4-ETHANAMINIUM INNER SALT |
| (a-S)-a-Carboxy-2,3-dihydro-N,N,N-trimethyl-2-thioxo-1H-imidazole-4-ethanaminium inner salt |
| [(1S)-2-hydroxy-2-oxo-1-[(2-thioxo-1,3-dihydroimidazol-4-yl)methyl]ethyl]-trimethyl-ammonium |
| 1H-Imidazole-4-ethanaminium, alpha-carboxy-2,3-dihydro-N,N,N-trimethyl-2-thioxo-, hydroxide, inner salt, (S)- |
| 1H-Imidazole-4-ethaniminium, alpha-carboxy-2,3-dihydro-N,N,N-trimethyl-2-thioxo-, hydroxide, inner salt, (S)- |
| Ammonium, (1-carboxy-2-(2-mercaptoimidazol-4-yl)ethyl)trimethyl-, hydroxide, inner salt, L-(+)- |
|
There are more than 10 synonyms. If you wish to see them all click here.
|