| 477-27-0 |
| O10-Demethylcolchicine |
| colchicein |
| N-Acetyl trimethylcolchicinic acid |
| NSC33411 |
| NSC 33411 |
| Coichiceine |
| O-Demethylcolchicine |
| Colchiceine (VAN) |
| O(sup 10)-Demethylcolchicine |
| UNII-HJ30158L57 |
| EINECS 207-512-5 |
| NSC-33411 |
| HJ30158L57 |
| NSC 123396 |
| NSC-123396 |
| BRN 2824078 |
| CHEMBL142588 |
| DTXSID60878565 |
| 4-14-00-00943 (Beilstein Handbook Reference) |
| NSC123396 |
| Benzo(a)heptalen-10(5H)-one, 7-acetamido-6,7-dihydro-9-hydroxy-1,2,3-trimethoxy- |
| Benzo(a)heptalen-9(5H)-one, 7-acetamido-6,7-dihydro-10-hydroxy-1,2,3-trimethoxy- |
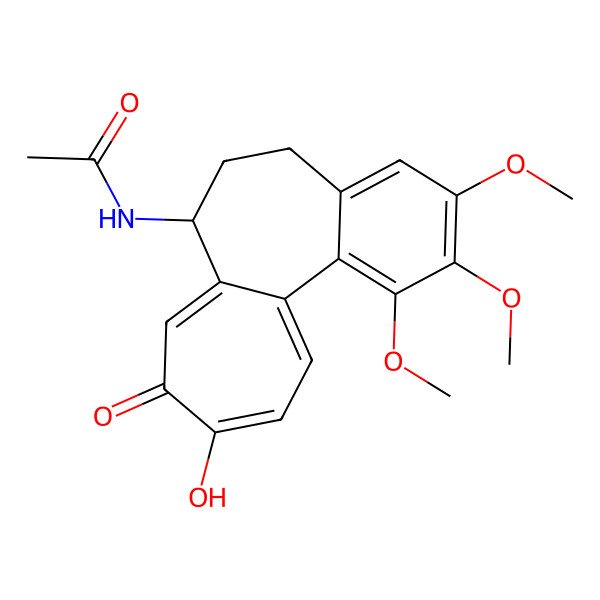
| N-[(7S)-10-hydroxy-1,2,3-trimethoxy-9-oxo-5,6,7,9-tetrahydrobenzo[a]heptalen-7-yl]acetamide |
| N-[(7S)-10-hydroxy-1,2,3-trimethoxy-9-oxo-6,7-dihydro-5H-benzo[a]heptalen-7-yl]acetamide |
| Acetamide, N-(5,6,7,9-tetrahydro-10-hydroxy-1,2,3-trimethoxy-9-oxobenzo(a)heptalen-7-yl)-, (S)- |
| ACETAMIDE, N-((7S)-5,6,7,9-TETRAHYDRO-10-HYDROXY-1,2,3-TRIMETHOXY-9-OXOBENZO(A)HEPTALEN-7-YL)- |
| N-((7S)-5,6,7,9-TETRAHYDRO-10-HYDROXY-1,2,3-TRIMETHOXY-9-OXOBENZO(A)HEPTALEN-7-YL)ACETAMIDE |
| 7-Acetamido-10-hydroxy-1,2,3-trimethoxy-6,7-dihydrobenzo(a)heptalen-9(5H)-one |
| WLN: L B677 MV&T&J CO1 DO1 EO1 JMV1 NQ |
| MFCD00598980 |
| Benzo[a]heptalen-9(5H)-one,7-dihydro-10-hydroxy-1,2,3-trimethoxy- |
| 7-Acetamido-10-hydroxy-1,3-trimethoxy-6,7-dihydrobenzo[a]heptalen-9(5H)-one |
| Acetamide, N-(5,6,7,9-tetrahydro-10-hydroxy-1,2,3-trimethoxy-9-oxobenzo[a]heptalen-7-yl)-, (S)- |
| Acetamide,6,7,9-tetrahydro-10-hydroxy-1,2,3-trimethoxy-9-oxobenzo[a]heptalen-7-yl)-, (S)- |
| N-{(7s)-10-hydroxy-1,2,3-trimethoxy-9-oxo-5,6,7,9-tetrahydrobenzo[a]heptalen-7-yl}acetamide |
| Spectrum_000357 |
| SpecPlus_000827 |
| COLCHICEINE [MI] |
| Spectrum2_000296 |
| Spectrum3_001161 |
| Spectrum4_000582 |
| Spectrum5_001013 |
| BSPBio_002741 |
| KBioGR_001044 |
| KBioSS_000837 |
| MLS002472977 |
| DivK1c_006923 |
| SCHEMBL177858 |
| SPECTRUM1800067 |
| SPBio_000232 |
| GTPL7525 |
| KBio1_001867 |
| KBio2_000837 |
| KBio2_003405 |
| KBio2_005973 |
| KBio3_002241 |
| CHEBI:183909 |
| DTXCID101016609 |
| HMS2205M17 |
| N-[(7S,12aRa)-10-Hydroxy-1,2,3-trimethoxy-9-oxo-5,6,7,9-tetrahydrobenzo[a]heptalen-7-yl]acetamide (Colchiceine) |
| TNP00282 |
| BDBM50157477 |
| CCG-39854 |
| AKOS004110611 |
| MS-2274 |
| SDCCGMLS-0066903.P001 |
| N-((7S)-10-hydroxy-1,2,3-trimethoxy-9-oxo-5,6,7-trihydrobenzo[d]heptalen-7-yl) acetamide |
| NCGC00017342-01 |
| NCGC00017342-02 |
| NCGC00017342-03 |
| NCGC00142518-01 |
| NCGC00178495-01 |
| NCI60_002925 |
| SMR001397084 |
| COLCHICINE IMPURITY F [EP IMPURITY] |
| Q27089242 |
| 7-Acetamido-10-hydroxy-1,2,3-trimethoxy-6,7-dihydrobenzo(a)heptalen-9(5H)-one, (S)- |
| N-[(7S)-10-hydroxy-1,2,3-trimethoxy-9-oxo-6,7-dihydro-5H-benzo[d]heptalen-7-yl]acetamide |
| (s)-n-(10-hydroxy-1,2,3-trimethoxy-9-oxo-5,6,7,9-tetrahydrobenzo[a]heptalen-7-yl)acetamide |
| 2SJ |
| N-((S)-10-Hydroxy-1,2,3-trimethoxy-9-oxo-5,6,7,9-tetrahydro-benzo[a]heptalen-7-yl)-acetamide |
| N-(10-Hydroxy-1,2,3-trimethoxy-9-oxo-5,6,7,9-tetrahydrobenzo[a]heptalen-7-yl)acetamide-, (S)- |
|
There are more than 10 synonyms. If you wish to see them all click here.
|