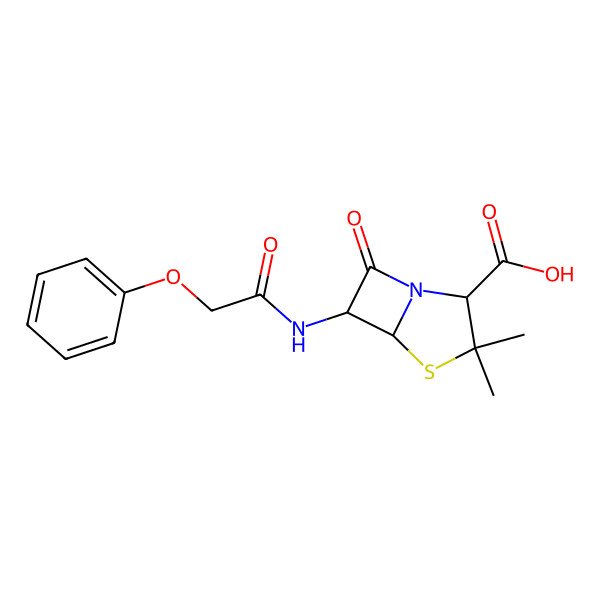
| Phenoxymethylpenicillin |
| Penicillin phenoxymethyl |
| Oracillin |
| 87-08-1 |
| Phenopenicillin |
| Phenomycilline |
| V-Cillin |
| Vebecillin |
| Fenospen |
| Distaquaine V |
| Phenoxymethyl penicillin |
| Meropenin |
| Calcipen |
| Fenacilin |
| Phenocillin |
| Phenoxymethylenepenicillinic acid |
| Stabicillin |
| Apopen |
| Oratren |
| Rocilin |
| Ospen |
| Compocillin V |
| Eskacillian V |
| Fenoximetilpenicilina |
| Beromycin |
| Eskacillin V |
| Phenoximethylpenicillinum |
| Phenoxymethylpenicillinum |
| Crystapen V |
| Pen-V |
| V-Tablopen |
| Acipen V |
| Pen-Oral |
| phenoxymethylpenicillinic acid |
| V-Cylina |
| V-Cyline |
| Phenoxymethylpenicilline |
| V-Cil |
| P-Mega-Tablinen |
| 6-Phenoxyacetamidopenicillanic acid |
| phenoxomethylpenicillin |
| Fenossimetilpenicillina |
| Pen-vee |
| Fenossimetilpenicillina [DCIT] |
| Fenoximetilpenicilina [INN-Spanish] |
| CCRIS 752 |
| CHEBI:27446 |
| HSDB 6314 |
| EINECS 201-722-0 |
| Phenoxymethylpenicilline [INN-French] |
| Phenoxymethylpenicillinum [INN-Latin] |
| UNII-Z61I075U2W |
| BRN 0096259 |
| (2S,5R,6R)-3,3-dimethyl-7-oxo-6-[(2-phenoxyacetyl)amino]-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid |
| PC Pen VK |
| Z61I075U2W |
| (2S,5R,6R)-3,3-Dimethyl-7-oxo-6-[(phenoxyacetyl)amino]-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid |
| Penicillanic acid, 6-phenoxyacetamido- |
| Penicillin V [USAN:USP] |
| Phenoxymethylpenicillin (INN) |
| Phenoxymethylpenicillin [INN] |
| 3,3-Dimethyl-7-oxo-6-((phenyloxyacetyl)amino)-4-thia-1-azabicyclo(3.2.0)heptane-2-carboxylic acid |
| (2S,5R,6R)-3,3-DIMETHYL-7-OXO-6-(2-PHENOXYACETAMIDO)-4-THIA-1- AZABICYCLO(3.2.0)HEPTANE-2-CARBOXYLIC ACID |
| (2S,5R,6R)-3,3-Dimethyl-7-oxo-6-(2-phenoxyacetamido)-4-thia-1-azabicyclo(3.2.0)heptane-2-carboxylic acid |
| (2S,5R,6R)-3,3-Dimethyl-7-oxo-6-(2-phenoxyacetamido)-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid |
| 4-Thia-1-azabicyclo(3.2.0)heptane-2-carboxylic acid, 3,3-dimethyl-7-oxo-6-((phenoxyacetyl)amino)-, (2S-(2alpha,5alpha,6beta))- |
| PNV |
| DTXSID3023429 |
| acipen-v |
| 4-27-00-05884 (Beilstein Handbook Reference) |
| 4-Thia-1-azabicyclo(3.2.0)heptane-2-carboxylic acid, 3,3-dimethyl-7-oxo-6-(2-phenoxyacetamido)- |
| PV |
| Fenoxymethylpenicillin |
| Penicillin V [USAN] |
| Penicillin, Phenoxymethyl |
| Penicillin V (USAN:USP) |
| Oracilline |
| Orocillin |
| PENICILLIN V (USP-RS) |
| PENICILLIN V [USP-RS] |
| Phenoxymethylpenicillin 100 microg/mL in Acetonitrile |
| Fenoximetilpenicilina (INN-Spanish) |
| 2,2-dimethyl-6beta-[(phenoxyacetyl)amino]penam-3alpha-carboxylic acid |
| PENICILLIN V (USP MONOGRAPH) |
| PENICILLIN V [USP MONOGRAPH] |
| Phenoxymethylpenicilline (INN-French) |
| Phenoxymethylpenicillinum (INN-Latin) |
| 3,3-dimethyl-6beta-[(phenoxyacetyl)amino]penam-2alpha-carboxylic acid (PIN) |
| PHENOXYMETHYLPENICILLIN (MART.) |
| PHENOXYMETHYLPENICILLIN [MART.] |
| Phenoxymethylpenicillinic acid potassium salt |
| (2S,5R,6R)-3,3-dimethyl-7-oxo-6-{[(phenyloxy)acetyl]amino}-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid |
| 4-Thia-1-azabicyclo(3.2.0)heptane-2-carboxylic acid, 3,3-dimethyl-7-oxo-6-((phenoxyacetyl)amino)-, (2S,5R,6R)- |
| PEN V |
| PHENOXYMETHYLPENICILLIN (EP MONOGRAPH) |
| PHENOXYMETHYLPENICILLIN [EP MONOGRAPH] |
| V-Cillin (TN) |
| Penicillin V (USP) |
| phenoxomethylpenicillanyl |
| phenoxymethylpenicillanyl |
| 2,2-dimethyl-6beta-((phenoxyacetyl)amino)penam-3alpha-carboxylic acid |
| 3,3-dimethyl-6beta-((phenoxyacetyl)amino)penam-2alpha-carboxylic acid (PIN) |
| (2S,5R,6R)-3,3-dimethyl-7-oxo-6-((phenoxyacetyl)amino)-4-thia-1-azabicyclo(3.2.0)heptane-2-carboxylic acid |
| PENICILLINV |
| PHENOSPEN |
| penicillin-v-potassium |
| Spectrum_001007 |
| PEN-VEE-ORAL |
| Spectrum2_000495 |
| Spectrum3_000543 |
| Spectrum4_000472 |
| Spectrum5_001409 |
| CHEMBL615 |
| Epitope ID:115011 |
| Epitope ID:116056 |
| PENICILLIN V [MI] |
| PENICILLIN V [HSDB] |
| SCHEMBL49223 |
| BSPBio_002185 |
| KBioGR_000944 |
| KBioSS_001487 |
| MLS001304105 |
| DivK1c_000779 |
| SPBio_000389 |
| DTXCID303429 |
| SCHEMBL22099709 |
| CHEBI:53706 |
| GTPL10920 |
| HY-B0975A |
| KBio1_000779 |
| KBio2_001487 |
| KBio2_004055 |
| KBio2_006623 |
| KBio3_001685 |
| J01CE02 |
| PENICILLIN, PHENOXYMETHYL- |
| BPLBGHOLXOTWMN-MBNYWOFBSA-N |
| NINDS_000779 |
| GLXC-25717 |
| PENICILLIN V [ORANGE BOOK] |
| 4-Thia-1-azabicyclo(3.2.0)heptane-2-carboxylic acid, 3,3-dimethyl-7-oxo-6-((phenoxyacetyl)amino)-, (2S-(2.alpha.,5.alpha.,6.beta.))- |
| BDBM50370584 |
| 6-(Phenoxyacetamido)penicillanic Acid |
| AKOS015969737 |
| DB00417 |
| IDI1_000779 |
| PHENOXYMETHYLPENICILLIN [WHO-DD] |
| PHENOXYMETHYLPENICILLIN [WHO-IP] |
| phenoxymethyl penicillinic acid*free acid |
| SMR000539431 |
| SBI-0051477.P003 |
| AB00514745 |
| CS-0013728 |
| Phenoxymethylpenicillin for system suitability |
| C08126 |
| D05411 |
| PHENOXYMETHYLPENICILLINUM [WHO-IP LATIN] |
| EN300-19735891 |
| Q422215 |
| W-109316 |
| BRD-K43966364-237-02-2 |
| BRD-K43966364-237-03-0 |
| 6beta-Phenoxyacetamido-2,2-dimethylpenam-3alpha-carboxylic acid |
| Phenoxymethylpenicillin, Antibiotic for Culture Media Use Only |
| Penicillin V, United States Pharmacopeia (USP) Reference Standard |
| Phenoxymethylpenicillin, European Pharmacopoeia (EP) Reference Standard |
| 4-THIA-1-AZABICYCLO(3.2.0)HEPTANE-2-CARBOXYLIC ACID, 3,3-DIMETHYL-7-OXO-6-((PHENOXYACETYL)AMINO)-, (2S-(2ALPHA,5ALPHA,6.BETA)).- |
|
There are more than 10 synonyms. If you wish to see them all click here.
|