Maysin
| Internal ID | 0fc03f5f-cc9f-4ef2-9f7d-87eab26900b1 |
| Taxonomy | Phenylpropanoids and polyketides > Flavonoids > Flavonoid glycosides > Flavonoid C-glycosides |
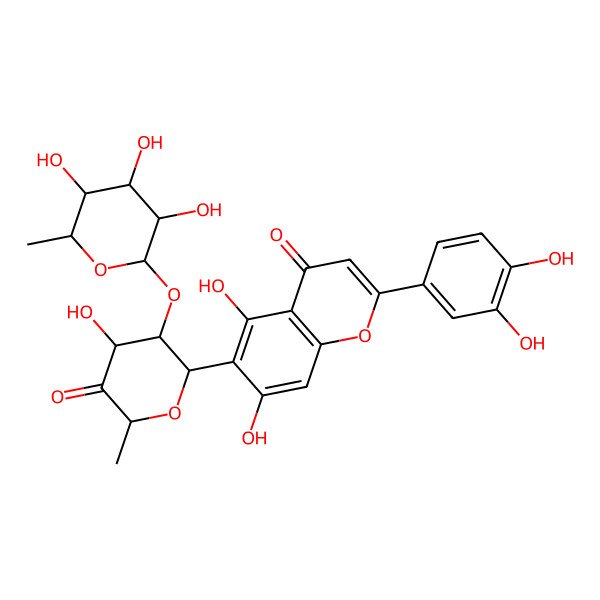
| IUPAC Name | 2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-6-[(2R,3S,4S,6S)-4-hydroxy-6-methyl-5-oxo-3-[(2R,3S,4S,5S,6R)-3,4,5-trihydroxy-6-methyloxan-2-yl]oxyoxan-2-yl]chromen-4-one |
| SMILES (Canonical) | |
| SMILES (Isomeric) | |
| InChI | InChI=1S/C27H28O14/c1-8-20(33)23(36)26(41-27-24(37)22(35)19(32)9(2)39-27)25(38-8)18-14(31)7-16-17(21(18)34)13(30)6-15(40-16)10-3-4-11(28)12(29)5-10/h3-9,19,22-29,31-32,34-37H,1-2H3/t8-,9+,19+,22-,23+,24-,25+,26-,27+/m0/s1 |
| InChI Key | GKLSYIMLZDYQBJ-CGOKOBCESA-N |
| Popularity | 31 references in papers |
| Molecular Formula | C27H28O14 |
| Molecular Weight | 576.50 g/mol |
| Exact Mass | 576.14790556 g/mol |
| Topological Polar Surface Area (TPSA) | 233.00 Ų |
| XlogP | -0.30 |
| Atomic LogP (AlogP) | -0.12 |
| H-Bond Acceptor | 14 |
| H-Bond Donor | 8 |
| Rotatable Bonds | 4 |
| 01O8RT377Y |
| CHEBI:70206 |
| 2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-6-((2R,4R)-4-hydroxy-6-methyl-5-oxo-3-((2S,4S,5R)-3,4,5-trihydroxy-6-methyloxan-2-yl)oxyoxan-2-yl)chromen-4-one |
| 2-(3,4-Dihydroxyphenyl)-5,7-dihydroxy-6-[(2R,4R)-4-hydroxy-6-methyl-5-oxo-3-[(2S,4S,5R)-3,4,5-trihydroxy-6-methyloxan-2-yl]oxyoxan-2-yl]chromen-4-one |
| 2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-6-[4-hydroxy-6-methyl-5-oxo-3-(3,4,5-trihydroxy-6-methyloxan-2-yl)oxyoxan-2-yl]chromen-4-one |
| RefChem:42475 |
| 2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-6-(4-hydroxy-6-methyl-5-oxo-3-(3,4,5-trihydroxy-6-methyloxan-2-yl)oxyoxan-2-yl)chromen-4-one |
| 3-Hexulose, 2,6-anhydro-1-deoxy-5-O-(6-deoxyhexopyranosyl)-6-C-(2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-4-oxo-4H-1-benzopyran-6-yl)- |
| 70255-49-1 |
| UNII-01O8RT377Y |
| There are more than 10 synonyms. If you wish to see them all click here. |
| Target | Value | Probability (raw) | Probability (%) |
|---|---|---|---|
| Human Intestinal Absorption | + | 0.7155 | 71.55% |
| Caco-2 | - | 0.8931 | 89.31% |
| Blood Brain Barrier | - | 0.9000 | 90.00% |
| Human oral bioavailability | - | 0.7571 | 75.71% |
| Subcellular localzation | Mitochondria | 0.7198 | 71.98% |
| OATP2B1 inhibitior | - | 0.5619 | 56.19% |
| OATP1B1 inhibitior | + | 0.8985 | 89.85% |
| OATP1B3 inhibitior | + | 0.9360 | 93.60% |
| MATE1 inhibitior | - | 0.8200 | 82.00% |
| OCT2 inhibitior | - | 0.9000 | 90.00% |
| BSEP inhibitior | + | 0.7756 | 77.56% |
| P-glycoprotein inhibitior | - | 0.5742 | 57.42% |
| P-glycoprotein substrate | - | 0.5373 | 53.73% |
| CYP3A4 substrate | + | 0.6502 | 65.02% |
| CYP2C9 substrate | - | 1.0000 | 100.00% |
| CYP2D6 substrate | - | 0.8669 | 86.69% |
| CYP3A4 inhibition | - | 0.8092 | 80.92% |
| CYP2C9 inhibition | - | 0.8456 | 84.56% |
| CYP2C19 inhibition | - | 0.9033 | 90.33% |
| CYP2D6 inhibition | - | 0.9601 | 96.01% |
| CYP1A2 inhibition | - | 0.8248 | 82.48% |
| CYP2C8 inhibition | + | 0.7732 | 77.32% |
| CYP inhibitory promiscuity | - | 0.7646 | 76.46% |
| UGT catelyzed | + | 0.7000 | 70.00% |
| Carcinogenicity (binary) | - | 1.0000 | 100.00% |
| Carcinogenicity (trinary) | Non-required | 0.6520 | 65.20% |
| Eye corrosion | - | 0.9908 | 99.08% |
| Eye irritation | - | 0.9049 | 90.49% |
| Skin irritation | - | 0.7203 | 72.03% |
| Skin corrosion | - | 0.9297 | 92.97% |
| Ames mutagenesis | + | 0.6363 | 63.63% |
| Human Ether-a-go-go-Related Gene inhibition | - | 0.3905 | 39.05% |
| Micronuclear | + | 0.9100 | 91.00% |
| Hepatotoxicity | - | 0.7125 | 71.25% |
| skin sensitisation | - | 0.9162 | 91.62% |
| Respiratory toxicity | + | 0.6000 | 60.00% |
| Reproductive toxicity | + | 0.7333 | 73.33% |
| Mitochondrial toxicity | + | 0.6375 | 63.75% |
| Nephrotoxicity | - | 0.9423 | 94.23% |
| Acute Oral Toxicity (c) | III | 0.5136 | 51.36% |
| Estrogen receptor binding | + | 0.8267 | 82.67% |
| Androgen receptor binding | + | 0.7613 | 76.13% |
| Thyroid receptor binding | + | 0.6012 | 60.12% |
| Glucocorticoid receptor binding | + | 0.7263 | 72.63% |
| Aromatase binding | + | 0.5368 | 53.68% |
| PPAR gamma | + | 0.7416 | 74.16% |
| Honey bee toxicity | - | 0.6100 | 61.00% |
| Biodegradation | - | 0.8500 | 85.00% |
| Crustacea aquatic toxicity | + | 0.6200 | 62.00% |
| Fish aquatic toxicity | + | 0.9691 | 96.91% |
Proven Targets:
| CHEMBL ID | UniProt ID | Name | Min activity | Assay type | Source |
|---|---|---|---|---|---|
| No proven targets yet! | |||||
Predicted Targets (via Super-PRED):
| CHEMBL ID | UniProt ID | Name | Probability | Model accuracy |
|---|---|---|---|---|
| CHEMBL5619 | P27695 | DNA-(apurinic or apyrimidinic site) lyase | 99.63% | 91.11% |
| CHEMBL1951 | P21397 | Monoamine oxidase A | 99.52% | 91.49% |
| CHEMBL2581 | P07339 | Cathepsin D | 98.01% | 98.95% |
| CHEMBL1806 | P11388 | DNA topoisomerase II alpha | 97.49% | 89.00% |
| CHEMBL1293255 | P15428 | 15-hydroxyprostaglandin dehydrogenase [NAD+] | 94.92% | 83.57% |
| CHEMBL2635 | P51452 | Dual specificity protein phosphatase 3 | 94.81% | 94.00% |
| CHEMBL4261 | Q16665 | Hypoxia-inducible factor 1 alpha | 92.67% | 85.14% |
| CHEMBL1860 | P10827 | Thyroid hormone receptor alpha | 91.91% | 99.15% |
| CHEMBL3108638 | O15164 | Transcription intermediary factor 1-alpha | 91.41% | 95.56% |
| CHEMBL1293249 | Q13887 | Kruppel-like factor 5 | 90.12% | 86.33% |
| CHEMBL3714130 | P46095 | G-protein coupled receptor 6 | 89.47% | 97.36% |
| CHEMBL3401 | O75469 | Pregnane X receptor | 86.25% | 94.73% |
| CHEMBL3475 | P05121 | Plasminogen activator inhibitor-1 | 86.04% | 83.00% |
| CHEMBL5845 | P23415 | Glycine receptor subunit alpha-1 | 85.15% | 90.71% |
| CHEMBL3038477 | P67870 | Casein kinase II alpha/beta | 83.80% | 99.23% |
| CHEMBL1293277 | O15118 | Niemann-Pick C1 protein | 81.40% | 81.11% |
| CHEMBL3194 | P02766 | Transthyretin | 80.86% | 90.71% |
| CHEMBL5608 | Q16288 | NT-3 growth factor receptor | 80.65% | 95.89% |
| PubChem | 70698181 |
| NPASS | NPC69260 |
| LOTUS | LTS0132611 |
| wikiData | Q27138546 |