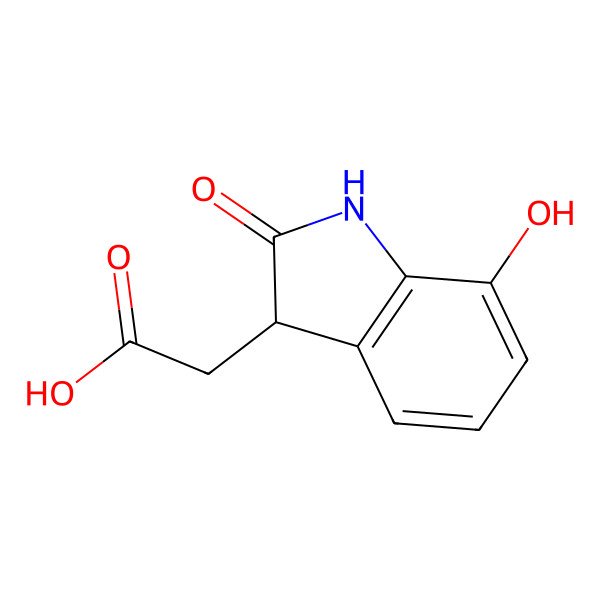
7-Hydroxy-2-oxindole-3-acetic acid
| Internal ID | e3fb20c2-0113-4314-a0de-59a15d89342f |
| Taxonomy | Organoheterocyclic compounds > Indoles and derivatives > Indolyl carboxylic acids and derivatives |
| IUPAC Name | 2-(7-hydroxy-2-oxo-1,3-dihydroindol-3-yl)acetic acid |
| SMILES (Canonical) | |
| SMILES (Isomeric) | |
| InChI | InChI=1S/C10H9NO4/c12-7-3-1-2-5-6(4-8(13)14)10(15)11-9(5)7/h1-3,6,12H,4H2,(H,11,15)(H,13,14) |
| InChI Key | RBODBEHKWJZUBG-UHFFFAOYSA-N |
| Popularity | 8 references in papers |
| Molecular Formula | C10H9NO4 |
| Molecular Weight | 207.18 g/mol |
| Exact Mass | 207.05315777 g/mol |
| Topological Polar Surface Area (TPSA) | 86.60 Ų |
| XlogP | -0.10 |
| Atomic LogP (AlogP) | 0.90 |
| H-Bond Acceptor | 3 |
| H-Bond Donor | 3 |
| Rotatable Bonds | 2 |
| SCHEMBL7651400 |
| Target | Value | Probability (raw) | Probability (%) |
|---|---|---|---|
| Human Intestinal Absorption | + | 0.9249 | 92.49% |
| Caco-2 | - | 0.8572 | 85.72% |
| Blood Brain Barrier | + | 0.6500 | 65.00% |
| Human oral bioavailability | + | 0.5857 | 58.57% |
| Subcellular localzation | Mitochondria | 0.7169 | 71.69% |
| OATP2B1 inhibitior | - | 1.0000 | 100.00% |
| OATP1B1 inhibitior | + | 0.9086 | 90.86% |
| OATP1B3 inhibitior | + | 0.9455 | 94.55% |
| MATE1 inhibitior | - | 0.8800 | 88.00% |
| OCT2 inhibitior | - | 0.9750 | 97.50% |
| BSEP inhibitior | - | 0.9388 | 93.88% |
| P-glycoprotein inhibitior | - | 0.9946 | 99.46% |
| P-glycoprotein substrate | - | 0.8207 | 82.07% |
| CYP3A4 substrate | - | 0.5379 | 53.79% |
| CYP2C9 substrate | - | 0.5779 | 57.79% |
| CYP2D6 substrate | - | 0.7747 | 77.47% |
| CYP3A4 inhibition | - | 0.9866 | 98.66% |
| CYP2C9 inhibition | - | 0.9271 | 92.71% |
| CYP2C19 inhibition | - | 0.9657 | 96.57% |
| CYP2D6 inhibition | - | 0.9484 | 94.84% |
| CYP1A2 inhibition | - | 0.8655 | 86.55% |
| CYP2C8 inhibition | - | 0.7423 | 74.23% |
| CYP inhibitory promiscuity | - | 0.9753 | 97.53% |
| UGT catelyzed | + | 0.7000 | 70.00% |
| Carcinogenicity (binary) | - | 0.8700 | 87.00% |
| Carcinogenicity (trinary) | Non-required | 0.6619 | 66.19% |
| Eye corrosion | - | 0.9931 | 99.31% |
| Eye irritation | + | 0.8948 | 89.48% |
| Skin irritation | - | 0.7980 | 79.80% |
| Skin corrosion | - | 0.9587 | 95.87% |
| Ames mutagenesis | - | 0.6300 | 63.00% |
| Human Ether-a-go-go-Related Gene inhibition | - | 0.8973 | 89.73% |
| Micronuclear | + | 0.8300 | 83.00% |
| Hepatotoxicity | + | 0.6711 | 67.11% |
| skin sensitisation | - | 0.8648 | 86.48% |
| Respiratory toxicity | + | 0.5889 | 58.89% |
| Reproductive toxicity | + | 0.8556 | 85.56% |
| Mitochondrial toxicity | + | 0.6375 | 63.75% |
| Nephrotoxicity | - | 0.6964 | 69.64% |
| Acute Oral Toxicity (c) | III | 0.6477 | 64.77% |
| Estrogen receptor binding | - | 0.8003 | 80.03% |
| Androgen receptor binding | + | 0.5481 | 54.81% |
| Thyroid receptor binding | - | 0.6927 | 69.27% |
| Glucocorticoid receptor binding | - | 0.6359 | 63.59% |
| Aromatase binding | - | 0.8863 | 88.63% |
| PPAR gamma | + | 0.6659 | 66.59% |
| Honey bee toxicity | - | 0.9724 | 97.24% |
| Biodegradation | - | 0.5750 | 57.50% |
| Crustacea aquatic toxicity | - | 0.7900 | 79.00% |
| Fish aquatic toxicity | + | 0.6441 | 64.41% |
Proven Targets:
| CHEMBL ID | UniProt ID | Name | Min activity | Assay type | Source |
|---|---|---|---|---|---|
| No proven targets yet! | |||||
Predicted Targets (via Super-PRED):
| CHEMBL ID | UniProt ID | Name | Probability | Model accuracy |
|---|---|---|---|---|
| CHEMBL5619 | P27695 | DNA-(apurinic or apyrimidinic site) lyase | 96.67% | 91.11% |
| CHEMBL2581 | P07339 | Cathepsin D | 96.31% | 98.95% |
| CHEMBL3108638 | O15164 | Transcription intermediary factor 1-alpha | 92.35% | 95.56% |
| CHEMBL3251 | P19838 | Nuclear factor NF-kappa-B p105 subunit | 90.69% | 96.09% |
| CHEMBL4203 | Q9HAZ1 | Dual specificity protein kinase CLK4 | 90.05% | 94.45% |
| CHEMBL2035 | P08912 | Muscarinic acetylcholine receptor M5 | 88.04% | 94.62% |
| CHEMBL3038477 | P67870 | Casein kinase II alpha/beta | 85.17% | 99.23% |
| CHEMBL4261 | Q16665 | Hypoxia-inducible factor 1 alpha | 84.34% | 85.14% |
| CHEMBL1860 | P10827 | Thyroid hormone receptor alpha | 82.17% | 99.15% |
| PubChem | 18398273 |
| LOTUS | LTS0226048 |
| wikiData | Q105233213 |